High-Volume Scanners
PRODUCTS
-
Expediting the customer journey from discovery all the way through commercial release.
-
Referenced in the new USP <1207> Chapter Guidance as a deterministic test method for container closure integrity testing.
-
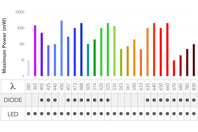
For decades, Melles Griot® Optical Systems have been at the forefront of illumination technology, partnering with researchers and instrument manufacturers to advance the study of biological samples.
-
The Thermo Scientific Sentinel 4000 Drop Through System, or Sentinel 4000D, featuring multiscan technology with up to five user-selectable frequencies of detection, provides improved sensitivity and detection of contaminants in drop through applications including granular and powdered food products, cosmetics, and chemicals.
-
A low‑risk, ultra‑efficient way to manage patient randomization and investigational product (IP) supply—fully unified within the TrialKit platform.
WHITE PAPERS AND CASE STUDIES
-
Enhancing Treatment Capacity For Simmons Foods
Learn how Simmons Foods resolved their wastewater treatment issues by replacing their aeration equipment, resulting in increased treatment capacity, improved removal rates, and reduced operating costs and energy consumption.
-
GeoTree - Delaware DOT Large Diameter Metal Plate Arch Rehabilitation With GeoSpray Geopolymer Mortar
Trenchless geopolymer technology provides a high-strength, structural solution for failing large-diameter arch culverts.This approach eliminates the need for costly bridge replacements, offering a 70-plus-year service life with minimal environmental impact and significantly faster project timelines.
-
A Feeder-Free Platform For Generating Mature Natural Killer Cells
NK cells represent a promising avenue for immunotherapy. Examine the advantages of NK cells in cellular immunotherapy and the importance of developing efficient activation and expansion platforms.
-
Leaks Go Acoustic: How Rogers Water Reduced Waste And Built Resiliency
Read how Rogers Water Utility (RWU) took action to mitigate constant leaks in their distribution system, which made it challenging to maintain volume in the eastern district’s water tower.
-
Turnkey Dismantling, Modification, And Relocation Of A Multi-Glove Isolator System
Strategic equipment relocation and retrofitting can reduce capital costs by fifty percent and save months in lead time. Learn how to modernize legacy systems while maintaining full GMP compliance.
-
Anaerobic Digestion Optimization System Provides Exceptional Improvements
Faced with soaring ammonia loads—especially during race weekends when 250,000+ fans descend on the town—the Speedway, Indiana Wastewater Treatment Plant was in urgent need of a solution.
-
Acoustic Inspection Helps Campbell River Optimize Inspection Program
In this case study, explore how incorporating the SL-RAT into their maintenance program has realized significant savings for the city of Campbell River.
-
If You Can't Win The Game, Change It
Explore how eData sharing is used in the biopharma market, enabling standardized, collaborative supply chains that reduce risks and enhance visibility while improving capacity planning.
-
From Months To Hours: MMC Medical's EDC Revolution
Learn how a specialized CRO reduced study build times by 95%. Discover strategies for template reusability and flexible resource scaling to accelerate early-stage medical device validation.
-
Pioneering Cancer Research Meets Unparalleled Veeva Vault Integration
A biotech company revolutionizing cancer treatment faced delays due to a complex, error-prone document review process. Discover how integrating an innovative strategy transformed their workflow.
-
Exploring Perfusion Technology For Virus Manufacturing Process Intensification
Perfusion cell culture technology supports high cell densities and product yields for viral vaccine manufacturing, offering a reliable way to produce high-quality cells at a commercial scale.
-
Leveraging High-Pressure Sterile Filtration For Highly Viscous Solutions
This research demonstrates the potential of high-pressure sterile filtration to enhance efficiency, reduce waste, and accelerate the development of innovative therapies.
NEWS
-
Falcon Oil & Gas Ltd ('Falcon') - Another Stellar IP60 Flow Test Result In The Beetaloo And 2025 Drilling Campaign Commences7/14/2025
Falcon Oil & Gas Ltd. (TSXV: FO, AIM: FOG) is pleased to announce that Shenandoah S2-2H ST1 (“SS-2H ST1”) achieved an average 60-day initial production (“IP60”) flow rate of 6.8 million cubic feet per day (“MMcf/d”) over 1,671-metres (5,483-foot) across a 35 stage stimulated horizontal within the Amungee Member B-Shale in the Beetaloo Sub-basin, Northern Territory, Australia, making it the highest IP60 result in the Beetaloo to date.
-
airSlate SignNow And Paperwise Partner To Redefine eSignature And Workflow Automation3/10/2025
airSlate, a global leader in electronic signature and document workflow automation, is excited to announce a new integration and reseller partnership with Paperwise, a premier provider of process automation and document management solutions.
-
Artificio Processes Documents In 80+ Languages, Enabling Multinational Corporations To Automate Global Operations12/9/2025
Artificio Products Inc., a leader in AI-powered intelligent document processing and enterprise workflow automation, today announced that its platform now processes documents in more than 80 languages, positioning the company as a comprehensive solution for multinational corporations managing cross-border document operations.
-
Crawford Technologies And Doceo Software Partner To Deliver Document Accessibility And Workflow Solutions Across Southern Europe8/27/2025
Crawford Technologies, a provider of innovative document solutions that streamline, improve and manage customer communications, expands the availability of its AccessibilityNow platform and award-winning workflow technology by establishing a partnership with Doceo Software, a provider of digital object management and document lifecycle automation, headquartered in Spain.
-
WHO Publishes 54 Pathogen Background Documents To Support Safe Water And Sanitation Systems6/21/2025
In a move to strengthen global efforts against waterborne disease, WHO has released 54 WASH-related pathogens new technical background documents — announced yesterday at a global water microbiology conference in the Netherlands — that capture the latest science on pathogens linked to drinking-water and sanitation systems.