Government
PRODUCTS
-
Building on the capabilities of the NP-RD10A benchtop tablet press (discussed in our previous article on USP <1062>), our services are further enhanced by the integration of the Presster™ compaction emulator. This state-of-the-art tool is designed to replicate the compaction dynamics of production tablet presses in a laboratory setting. It allows for tangible fine-tuning of the compression cycle, providing an accurate depiction of how a tablting formulation will behave under commercial manufacturing conditions. Using the Presster, Natoli Scientific can simulate full-scale production, permitting us to forecast problems before they escalate to costly production delays. Our experts can optimize the tablet design for our clients, ensuring a smooth transition from laboratory to production while adhering to the rigorous standards set by the industry including the current USP <1062> chapter.
-
Keeping your community safe in the face of water quality concerns is a complicated task. With the help of waterCAST WQ, you can track your water quality compliance and make informed decisions to safeguard your community.
-
A successful clinical trial starts with an efficient protocol review.
With deep roots in academic research and clinical medicine, BRANY IRB is uniquely positioned to meet your cost and speed requirements. Get your clinical trial to the start line while ensuring participant health and safety.
-
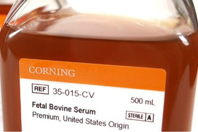
Fetal Bovine Serum (FBS) is a time tested, universal supplement used in cell culture. It provides many compounds that have been shown to satisfy specific metabolic requirements for the culture of cells, including hormones, growth factors, cell attachment factors, pH buffer, protease inhibitors, vitamins, minerals, lipids, and a variety of undefined components.
-
Levitating transport technology is redefining aseptic production. Reduce your footprint and carbon impact while streamlining the fill-finish process with a simplified, frictionless workflow.
WHITE PAPERS AND CASE STUDIES
-
How The Accelerator™ Drug Development Model Enhances eNPV
Discover how an integrated CDMO and CRO model can accelerate development timelines, add up to $62.9 million in value, and help you maximize long-term pipeline success with a single, streamlined partner.
-
Pittsburgh Water Sets New Standard With Customer Request Sampling To Support Lead Service Line Replacement
Discover how 120Water helped Pittsburgh Water reduce their turnaround times, streamline sampling workflows, and save hours of work. Learn how you can also use sampling to support your replacement and community engagement programs with their case study.
-
Deal-Making Volume Slowed In 2023
This report provides an overview of alliance, merger and acquisition, and financing deal activities across the global biopharma, medical device, and in vitro diagnostics industries in 2023, as reported by Biomedtracker.
-
Rethinking Clinical Operations: A Smarter, Faster Playbook For Trial Success
The CRO-sponsor partnership is now strategic, addressing communication gaps and fragmented systems to improve efficiency. Joint governance, real-time tracking, and unified technology accelerate complex trial execution.
-
From Months To Hours: MMC Medical's EDC Revolution
Learn how a specialized CRO reduced study build times by 95%. Discover strategies for template reusability and flexible resource scaling to accelerate early-stage medical device validation.
-
Meet Regulation Requirements With RTU Vials And Cartridges
Sterile manufacturing remains vital, with resilient supply chains preventing shortages. Industry and regulators prioritize integrity as demand surges, making it the fastest-growing pharmaceutical segment.
-
Preserving Pressure, Protecting Service: Upgrading Hydrant Infrastructure In Kent County
When Kent County Water Authority faced the urgent replacement of a 94-year-old hydrant tied to a 1931 cast iron main, they were determined to maintain fire protection and avoid customer disruption.
-
Incremental Adoption Of A Modular Operations Platform
Discover the potential of a modular operations platform that scales the benefits of digitalization and aids organizations in implementing a unified data strategy to realize the full potential of smart manufacturing.
-
How LIMS Supports QC Scaling At Forge Biologics
Modernizing QC workflows with cloud-based systems eliminates paper inefficiencies, reduces errors, and accelerates reporting. Discover how streamlined processes improve compliance and enhance data visibility.
-
Does Asset Health Monitoring Have A Role In The Water Sector?
See why collaboration with trusted vendors is crucial for deploying reliable, adaptable, and sustainable asset health solutions in the evolving water sector.
-
What Clinical Trial Decision Makers Are Prioritizing In 2025
In 2025, clinical trial operations are undergoing a transformative shift driven by the need for speed, cost-efficiency, and patient-centered approaches.
-
Severn Trent Exceeds Leak Reduction Targets With Innovative Itron Water Solutions
Severn Trent’s smart metering program, powered by Itron’s technology, is transforming water conservation—reducing leaks by 6 million liters, cutting costs, and empowering customers to use water more wisely.
NEWS
-
AtkinsRéalis Welcomes UK Infrastructure Pipeline7/17/2025
The UK Government has today launched a new Infrastructure Pipeline and interactive online tool providing real-time updates on more than 780 planned infrastructure projects across the UK.
-
New Open-Access Book Edited By Council Member Manny Teodoro Examines The Future Of U.S. Drinking Water3/24/2026
Fifty years after the passage of the Safe Drinking Water Act (SDWA), a new open-access book looks ahead to what it will take to protect America’s drinking water in the decades to come.
-
Philippines Launches National Plastic Action Partnership To Tackle Plastic Waste And Advance Circular Economy3/9/2025
The Philippines took a significant step forward in addressing its plastic waste crisis with the launch of the National Plastic Action Partnership (NPAP), a multi-stakeholder platform designed to foster collaboration across sectors and accelerate the country’s transition to a circular economy.
-
Crawford Technologies And Doceo Software Partner To Deliver Document Accessibility And Workflow Solutions Across Southern Europe8/27/2025
Crawford Technologies, a provider of innovative document solutions that streamline, improve and manage customer communications, expands the availability of its AccessibilityNow platform and award-winning workflow technology by establishing a partnership with Doceo Software, a provider of digital object management and document lifecycle automation, headquartered in Spain.
-
R2 Recycling Sets The Standard For E-Waste Recycling Excellence In Paterson3/12/2025
R2 Recycling is proud to announce its unwavering commitment to providing top-quality e-waste recycling solutions to businesses, schools, and households across Paterson, New Jersey.