Project Management & Site Enablement
Set sites up for success with protocol-specific training and hands-on guidance that drives confident technology use, faster adoption, and fewer in-study issues.

Digital health technologies have the potential to transform clinical trials, yet without careful planning and operational support, their implementation can create complexity, increase site burden, and jeopardize data quality.
Backed by over 20 years of experience across nearly 300 industry-sponsored trials, Ametris’ Digital Health Operations team delivers end-to-end trial enablement services to ensure seamless technology deployment, reduce site burden, and capture reliable,
Adherence Monitoring Solutions
Maximize the value of your DHT investment with the most advanced adherence monitoring tools in the industry.
Participant non-adherence and missing data are among the most common causes of protocol deviations, increased development costs, and delayed timelines. Ametris’ integrated ActiTrack adherence monitoring solutions help study teams identify issues early, intervene appropriately, and preserve data quality, whether you need high-level visibility or near real-time action.

ActiTrack
Designed for teams that need clear visibility into adherence trends without disrupting study workflows, ActiTrack Adherence Monitoring helps you spot emerging issues and focus support where it’s needed most.
Ideal for: Studies needing clear adherence oversight and trend analysis
Ensure your digital health technology assessments deliver reliable, high-quality data with expert operational support from study planning through closeout and beyond.
Project Management & Site Enablement
Set sites up for success with protocol-specific training and hands-on guidance that drives confident technology use, faster adoption, and fewer in-study issues.
Logistics & Device Management
Eliminate the complexity of device distribution and tracking. We handle shipping to sites, inventory oversight, and replacements to keep studies running smoothly and on schedule.
In-Trial Data Monitoring & Adherence Insights
Protect data integrity with ongoing in-trial screening and actionable reporting that helps teams quickly identify and address adherence or data quality risks.
Data Exports & Transfers
Simplify downstream data management with exports and transfer formats tailored to your sponsor or CRO requirements, enabling easier dataset alignment and faster study results.
Participant Profile Reporting
Enhance patient-centricity by providing participants with clear, end-of-study summaries that reinforce engagement and study value.
Data Archiving
Close out studies with confidence through secure, seamless data archiving aligned with regulatory and retention requirements.
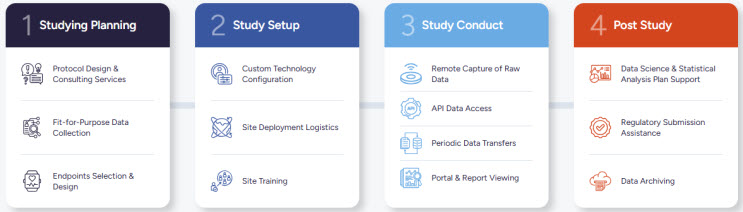
Our Process
End-to-End Trial Enablement Support