Post-Marketing Studies And Real-World Evidence Clinical Trials
Your trusted partner for decentralized post-marketing surveillance clinical trials and real-world evidence collection. Point-of-need accessibility. Proven compliance.

Traditional fixed-location sites face persistent challenges in post-marketing surveillance and post-approval studies. Limited patient access slows real-world data capture, while travel burden and scheduling constraints reduce retention and compromise patient safety monitoring. High operational costs and limited flexibility also make it difficult to generate evidence-based insights across diverse populations, which regulatory bodies such as the U.S. Food and Drug Administration (FDA) increasingly expect for ongoing safety studies.
|
Low Patient Compliance Patients often skip follow-up visits due to travel inconvenience, reducing adherence, study consistency, and overall long-term data reliability. |
Limited Real-World Data Restricted geographic reach limits diverse population representation, weakening study validity, generalizability, and real-world insight into treatment response, side effects, and product safety. |
High Dropout Rates Traditional trials experience 30–40% dropout in long-term studies, reducing statistical power, delaying timelines, and putting post-marketing commitments for new drugs at risk. |
Delayed Data Collection |
Why Choose 20/20 Onsite for Post-Marketing Surveillance Clinical Trials?
Post-marketing surveillance clinical trials are essential for monitoring drug safety and efficacy after FDA approval. At 20/20 Onsite, we revolutionize traditional post-marketing studies by bringing comprehensive ophthalmic assessments directly to patients' preferred locations, eliminating barriers that typically reduce participation and data quality in real-world evidence studies.
Our decentralized approach addresses critical challenges facing pharmaceutical sponsors: low patient compliance, limited geographic reach, and high dropout rates that compromise study integrity. By delivering point-of-need clinical services to homes, workplaces, and community centers, we ensure superior patient retention while capturing authentic, real-world data.
This innovative model enables sponsors to meet post-marketing commitments more efficiently, reduce operational costs, and generate higher-quality real-world evidence that supports regulatory submissions and market access strategies.

Our Post-Marketing Surveillance Services
Our comprehensive approach supports FDA post-marketing surveillance requirements, with Phase IV clinical trial services and post-approval drug safety monitoring that help sponsors meet market authorization obligations across diverse therapeutic areas.
Study, Design, & Planning
- Target population analysis for real-world evidence studies
- FDA post-marketing requirements and commitments (PMR/PMC) planning
- Post-marketing surveillance clinical trials protocol development
- Site selection for decentralized post-marketing surveillance
Implementation & Data Collection
- Multi-site post-market clinical research execution
- Point-of-care surveillance and real-world data collection
- Digital pharmacovigilance studies platforms
- Flexible assessment scheduling for clinical trial post-marketing studies
Data Reporting
- Ongoing real-world evidence studies monitoring
- Post-approval clinical studies data reporting
- Regulatory reporting and safety signal detection
- Long-term outcome assessment and intervention
- FDA Sentinel Initiative safety monitoring compliance
Study Design & Planning
Clinical Trial Post-Marketing Studies with Comprehensive Protocol Development and Strategic Planning
Effective real-world evidence studies begin with a thorough study design and planning, yet many post-marketing surveillance clinical trials face challenges due to inadequate protocol development and population analysis.
Traditional planning approaches often overlook critical factors like real-world patient populations, geographic diversity, and regulatory compliance requirements that impact decentralized post-marketing surveillance success.
How Our Clinical Research Support Services Help?
20/20 Onsite provides comprehensive study design and planning services that establish the foundation for successful post-market clinical research through detailed target population analysis and feasibility studies for real-world evidence studies.
Our experienced team develops optimized clinical trial post-marketing studies strategies tailored to your specific post-approval requirements, including site selection for geographic diversity and regulatory compliance assessment for all post-marketing surveillance materials.
Implementation & Data Collection
Post-Marketing Surveillance with Multi-Site Execution and Real-World Data Capture
During the implementation phase, post-market clinical research requires efficient data collection across diverse patient populations in real-world settings, yet many studies struggle with traditional site limitations and fragmented data collection approaches.
Traditional collection methods often rely on fixed-site approaches that miss key patient segments, particularly when real-world evidence studies require specialized populations or point-of-care surveillance capabilities.
How Our Clinical Research Support Services Help?
20/20 Onsite executes comprehensive multi-site clinical trial post-marketing studies that combine community outreach, physician networks, and digital pharmacovigilance studies platforms for maximum real-world data collection efficiency.
Our post-marketing surveillance clinical trials team coordinates point-of-care surveillance and flexible assessment scheduling, bringing decentralized post-marketing surveillance directly to participants through mobile capabilities and community partnerships.
Data Reporting
Post-Marketing Surveillance with Comprehensive Regulatory Reporting
Maintaining data integrity and regulatory compliance throughout post-marketing surveillance clinical trials is critical for safety monitoring and regulatory requirements, yet data reporting challenges remain significant across all real-world evidence studies.
Traditional approaches often fail to address the complex data integration and safety signal detection requirements that are essential for effective pharmacovigilance studies and post-approval clinical studies.
How Our Clinical Research Support Services Help?
20/20 Onsite delivers ongoing real-world evidence studies monitoring through comprehensive safety signal detection and regulatory reporting that keeps post-market clinical research compliant throughout the study.
Our clinical trial post-marketing studies specialists provide dedicated support and regulatory reporting coordination, with point-of-care surveillance capabilities that ensure continuous data quality and safety monitoring.
Our post-marketing surveillance capabilities include comprehensive regulatory support for FDA compliance
We provide pharmacovigilance outsourcing post-approval with expertise in FDA post-marketing surveillance requirements.
|
Struggling With Post-Marketing Patient Compliance? Maintain patient engagement in long-term post-marketing surveillance clinical trials by making follow-up ophthalmic assessments more convenient and accessible through point-of-need services. |
Need Support with Real-World Evidence Collection? Ensure patients participating in post-marketing studies who require ongoing safety monitoring stay compliant with their scheduled follow-up appointments and data collection requirements. |
Trying to Meet FDA Post-Marketing Commitments? We ensure that the post-marketing surveillance experience delivers comprehensive regulatory compliance by providing specialized clinical personnel to conduct these assessments with appropriate documentation and accessibility. |
Our 5 Steps Post-Marketing Surveillance Excellence Framework
Industries We Serve for Post-Marketing Studies
|
Pharmaceutical Companies
|
Medical Device Manufacturers
|
Biotechnology Firms
|
Benefits of Our Post-Marketing Study Services
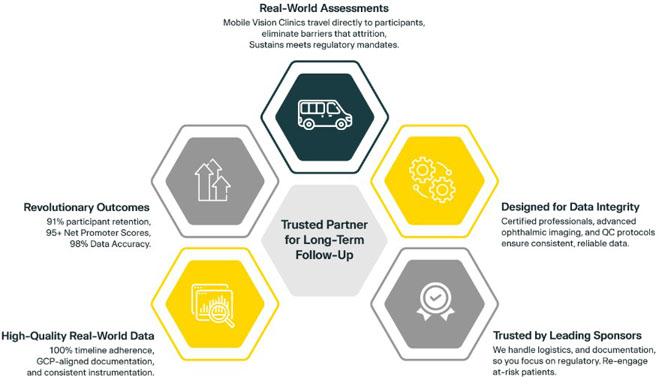
1. Trusted Partner for Long-Term Follow-Up
20/20 Onsite handles all necessary logistics for post-marketing surveillance clinical trials point-of-need assessments, including travel, setup, exams, data submission and real-time technical support. With over 85,000 eye exams performed, 40+ clinical trials supported, and an average of 11+ years of ophthalmic experience per clinical professional, we deliver long-term ocular follow-up anywhere in the U.S.
2. Real-World Assessments Without the Drop-Off
Our Mobile Vision Clinics travel directly to participants for real-world evidence studies, eliminating barriers that cause attrition. Post-marketing surveillance clinical trials benefit from our national fleet, bringing protocol-specific eye exams wherever participants are located, sustaining engagement, and meeting regulatory mandates without sacrificing convenience.
3. Designed for Data Integrity in the Real World
Our certified decentralized post-marketing surveillance professionals ensure high-quality assessments with advanced ophthalmic imaging and diagnostic tools, integrating visual acuity, IOP, OCT, fundus photography, and slit lamp exams. Centralized image upload and built-in quality control protocols deliver consistent, reliable clinical trial post-marketing studies data.
4. Trusted by Leading Sponsors for Long-Term Follow-Up
We handle logistics, scheduling, and documentation for post-market clinical research, so you focus on regulatory compliance. Our team collaborates to identify participant access gaps, re-engage at-risk patients, conduct in-home assessments, and deliver scalable solutions, keeping real-world evidence studies on track.
5. High-Quality Real-World Data Collection and Reporting
With 98% data accuracy and 100% compliance with the clinical trials timeline, we deliver reliable results that meet regulatory standards. Our GCP-aligned documentation and consistent instrumentation ensure no variability due to local providers or logistical challenges, just repeatable data every time.
Why Do Post-Marketing Surveillance Strategies Work?
20/20 Onsite combines convenience, expertise, and technology to improve post-marketing surveillance clinical trials, real-world evidence studies, participant retention, and data integrity, driving successful, patient-centric post-marketing follow-up studies. From eliminating participant travel barriers to achieving retention rates as high as 91% (vs industry standard 70%), our point-of-need assessment methodology delivers measurable results that fulfill regulatory mandates while potentially reducing post-marketing surveillance operational costs.
Revolutionary Post-Marketing Surveillance Outcomes:
- Advanced engagement protocols maintain 91% participant retention rates
- Consistent instrumentation ensures data integrity across all timepoints
- Point-of-need accessibility achieves 95+ Net Promoter Scores
- GCP-aligned documentation maintains 98% regulatory compliance
