Manufacturing Solutions
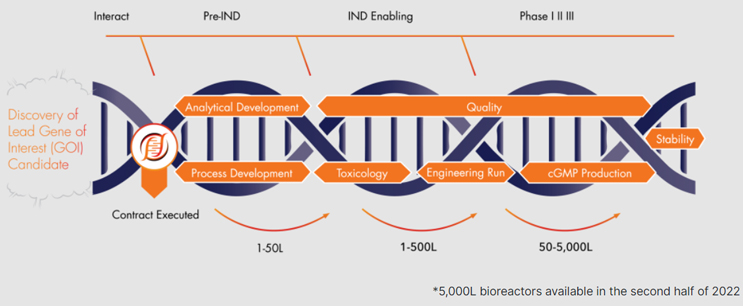
The foundation of our business is "The Hearth," a 200,000+ ft2 custom-designed, cGMP facility, dedicated to AAV viral vector manufacturing. We offer end-to-end manufacturing and development services to accelerate gene therapy programs from preclinical, through clinical, and commercial manufacturing.
Blaze Vector Production
Turnkey, scalable, premier research-grade AAV production
- Our proprietary Ignition Cells™ that have a clear, well-documented history and are optimized for scalable suspension growth
- Established process for many standard AAV serotypes
- Upfront communication and pricing
- Fast turnaround time
Process & Analytical Development
End-to-end capabilities from research to cGMP, reducing the need for bridging studies.
- Experienced team able to support process development and scale up
- In-house analytical assay development and performance to support cGMP-readiness
- Process development to manufacture novel engineered capsids
cGMP Production
Fully disposable, closed system processing.
- Flexible scheduling and maximization of capacity
- Consistent process and product flow for reduction of variation
