Healthcare
PRODUCTS
-
Our team of experts are equipped to guide you through every step of any Inspection, especially Pre-Approval Inspections, due to the nature of their criticality.
-
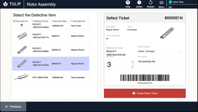
Track and manage defects and non-conformances – such as SOP deviations or missing data – from identification to resolution.
-
We support you in the process of optimally implementing complex Manufacturing Execution Systems. In addition to the creation of Master Batch Records (MBRs) and Electronic Batch Recording (EBR), including classic data acquisition and preparation, as well as all other processes that have immediate effects on the manufacturing process.
-
Evolve from paper to life sciences digital operations quickly and simply, paving the fastest path to fewer deviations and faster batch releases with data-driven manufacturing.
-
Whether you’re conducting an oncology trial, a neurology study, or imaging-based endpoint research, TrialKit’s flexible imaging capabilities can meet your needs.
WHITE PAPERS AND CASE STUDIES
-
Operational Success By Strategic Investment
Explore how infrastructure, equipment strategy, and advanced technologies shape a more reliable path for scaling sterile injectable programs.
-
Mobile App And Wearable Integration Collects Longitudinal Data
Get an overview of SISCAPA's utilization of eTechnologies, specifically ePRO, and how their partnership with CDS accurately demonstrates the improvement of data collection and analysis in the biotechnology industry.
-
Leveraging Life Sciences Data For Intelligent Decision-Making
Industry 4.0 drives life sciences to prioritize data-driven decision-making by integrating advanced technologies in QMS. Discover how modern data management can improve compliance.
-
The Business Case For EHR eSource In Clinical Trials
Examine how manual data collection challenges prompted the adoption of an eSource solution that streamlined data entry, saving time and costs, reducing errors, and accelerating a multicenter phase 2 trial.
-
Digitalizing Batch Records In Pharmaceutical Production
Pharma manufacturing embraces cutting-edge automation, yet outdated paper-based documentation hinders efficiency and delays batch releases. See how digital solutions can transform compliance.
-
How Medidata Link Is Supporting Moderna's Clinical Trial Real-World Data Linkage Initiatives
Discover how Medidata Link empowers Moderna's clinical trial RWD linkage initiatives, enhancing insights into treatment outcomes while minimizing administrative burdens.
-
Taking Research In-House With CRIO: SciTech's Multi-Center Phase 1 Study
Explore the success story of an early-stage clinical pharmaceutical company that effectively navigated funding constraints when conducting its Phase 1 study without a third-party CRO.
-
Digital Standardization: Accelerating Gene Therapy Tech Transfer With Confidence And Compliance
Fragmented, manual data practices slow tech transfer and increase risk; centralized digital CMC systems standardize workflows, improve transparency, and accelerate onboarding while maintaining compliance.
-
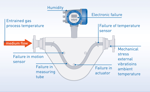
OPTICHECK Technology Built-In – Diagnostics, Verification, And Functional Safety With OPTIMASS Coriolis Mass Flow Meters
State-of-the-art measuring devices such as OPTIMASS Coriolis mass flow meters combine robust measuring principles and powerful electronics to produce a wide range of readings and device-specific data. OPTICHECK technology built-in leverages the features of the devices to translate their available comprehensive measuring system data into sophisticated diagnostics information for plant personnel.
-
Overcoming Analytical Bottlenecks In Oligonucleotide Drug Development With Automation
Growing oligonucleotide complexity is straining traditional analytical workflows. See how automated LC‑UV‑MS processing offers a scalable way to boost throughput and reduce manual effort.
-
Providing Enhanced Manufacturing Data To Contracting Companies
The life sciences sector is projected to reach $220 billion by 2030. Examine the growth of CDMOs and CMOs, emphasizing the need for digitalization to meet client demands and regulatory standards.
-
Seamless Onshoring From China-Based CDMO
Successfully mitigate geopolitical risk and maintain supply continuity. Learn how a large-scale manufacturing process was rapidly requalified in eight months to secure a clinical-stage biologic program.
NEWS
-
Samtec To Exhibit At OFC 2026, Participate In OIF Interoperability Demo3/10/2026
Samtec, Inc., the service leader in the connector industry, will be demonstrating next-generation, high-performance optical and copper interconnect solutions in South Hall booth #1101 at the upcoming OFC exhibition (March 17-19 at Los Angeles Convention Center, Los Angeles, CA).
-
CEA-Leti Builds Pathways To Improved Chemical Detection, High-Speed Communication And LIDAR Performance Via Integrated Optics-On-Silicon Advances1/30/2025
CEA-Leti scientists presented three papers at Photonics West 2025 detailing the institute’s latest R&D successes to improve chemical detection, high-speed communication and LIDAR performance with integrated optics on silicon.
-
Mobisoft Infotech Is Digitizing Global Transport Operations With Intelligent Fleet Technology3/17/2026
As global supply chains grow increasingly complex, customer expectations for faster and more reliable deliveries continue to rise. Enterprises across every sector are rethinking how they run transportation from the ground up.
-
allGeo Strengthens Homecare Agency Operations With Its End-To-End Electronic Visit Verification Platform4/4/2025
allGeo, a service by Abaqus Inc. and a leading field service automation platform, offers robust capabilities in its Electronic Visit Verification (EVV) system, built to help homecare and personal care agencies drive operational excellence.
-
Predictiv AI Expands Into Hardware With Shiftmatics Platform And Secures Initial Client Order4/14/2026
Predictiv AI Inc. (the “Company” or “Predictiv AI”) announces the completion of Phase 1 of its Shiftmatics hardware platform and the receipt of its first client order. This milestone marks the Company’s expansion into hardware-enabled AI infrastructure, supporting real-time intelligence and future autonomous fleet operations.